|
3/10/2024 0 Comments Bohrs model of the atom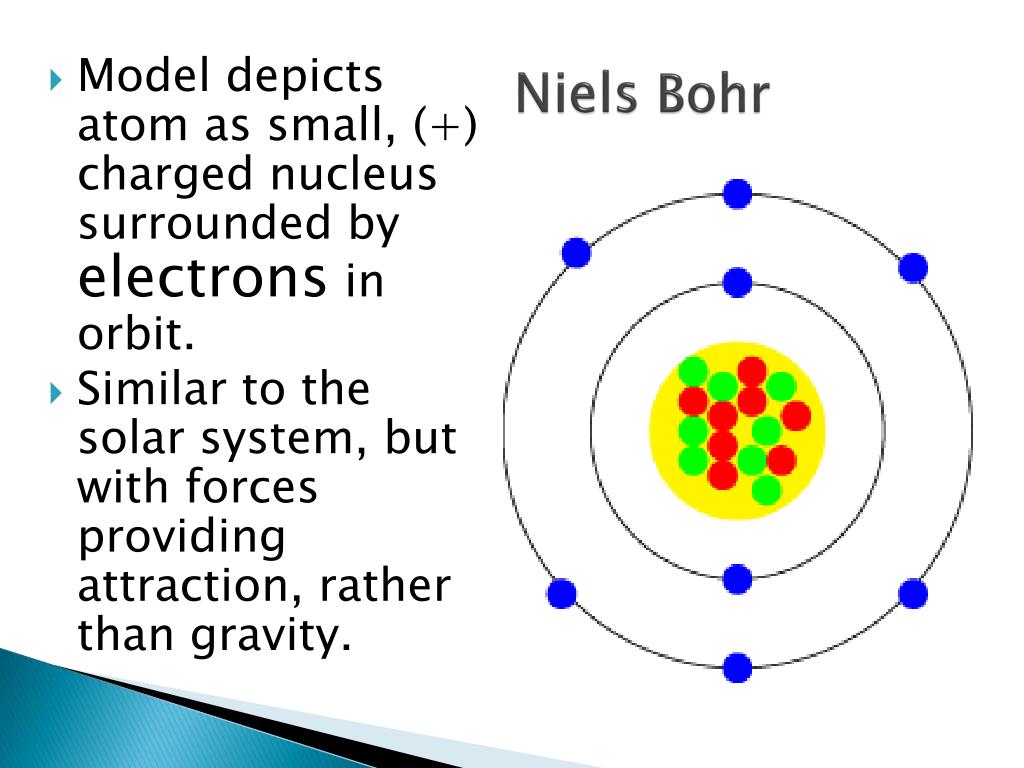 
As Bohr had noticed, the radius of the n 1 orbit is approximately the same size as an atom. Basically, if an electron with x amount of energy goes to a lower state of energy and loses y energy, then a photon with energy y is emitted by the atom and either becomes light or some other form of radiation. In Bohr’s model, radius an of the orbit n is given by the formula an h2n2 0 / 2, where 0 is the electric constant. Bohr agreed with classical theory that light has a wave-particle duality (meaning that it is made of both electromagnetic waves and particles called photons), but he said that light was given off when an electron in a hydrogen atom jumps from a higher energy level to a lower one. An early model of the atom was developed in 1913 by Danish scientist Niels Bohr (18851962). This means that if an electron jumps from one energy level to the next, it will never be in between energy levels, but will instantaneously be transported from one level to the other.īohr's model is also useful because it explains the cause of light. The early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. 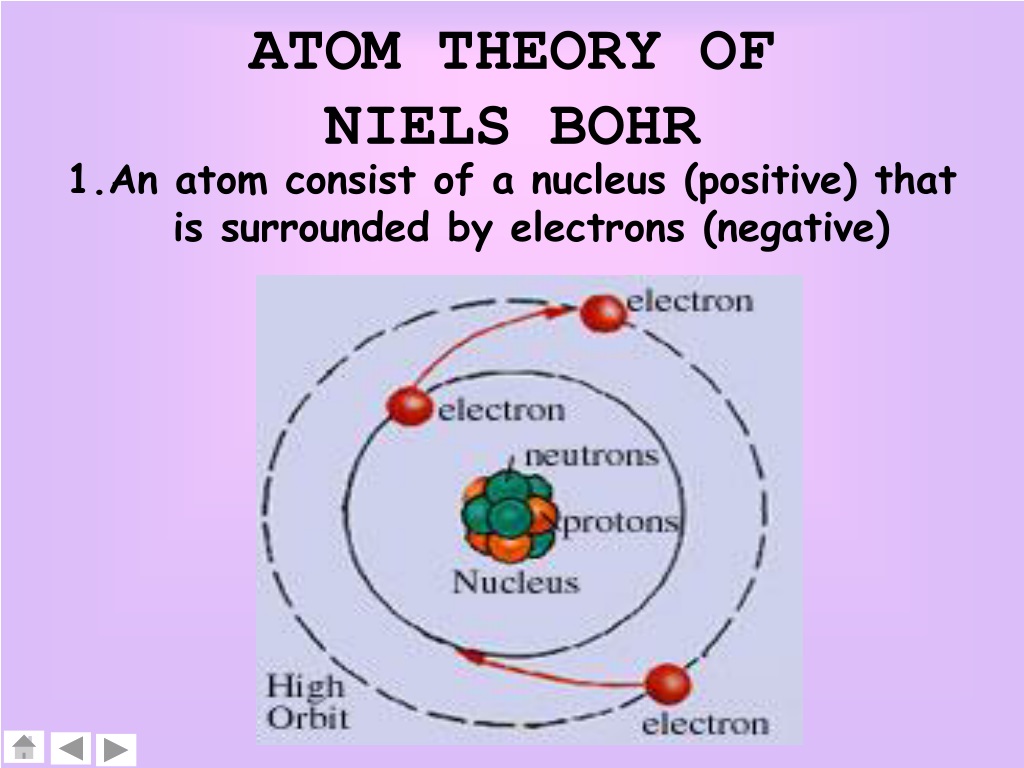
This formula says that an electron will only be found on the energy levels predicted by the formula, and not anywhere in between. Where h is Planck's constant, π is pi, and n is the multiples (1,2,3.). The Bohr model says that the angular momentum of an electron within a hydrogen atom can only be integer multiples of a certain number. Bohr's model is extremely useful in describing the hydrogen emission spectrum.Įlectrons Energy Levels The Bohr model gives the specific energy levels of an electron (n=1,2,3) Up to 2 electrons orbit in the first energy level. The different orbit distances are called energy levels. In the Bohr model of the atom: Electrons orbit the nucleus at different distances. He used the nuclear model to create his model. These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells. In 1913 the Danish Physicist, Niels Bohr, came up with an improved model of the atom. Bohr's model is useful only when discussing the behavior of a hydrogen atom. The Bohr model shows the atom as a central nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus (Figure 1). In quantum mechanics, a branch of physics, the Bohr model is the model of the atom proposed by Niels Bohr.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
